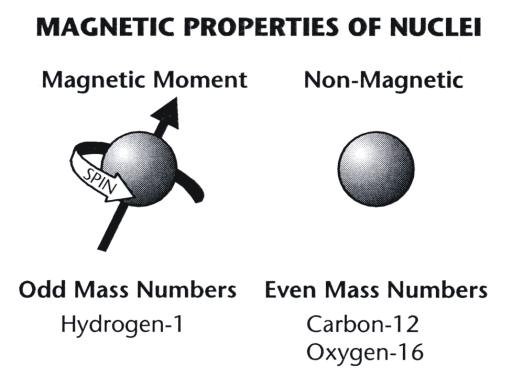
10 EXAMPLE Magnetic dipole moment of a revolving electron An electron revolving in an orbit of radius 0.5 À in a hydrogen atom executes 10 revolutions per second. Find the magnetic moment

The electron in the hydrogen atom is moving with a speed of 2.3 x 10^6 m/s in an orbit of radius 0.53 A. Calculate the magnetic moment of the revolving electron. - Sahay LMS
In a hydrogen atom, an electron of charge revolves in an orbit of radius r with speed v. What is the magnitude of the resulting magnetic moment of the electron? - Quora

An electron in the ground state of hydrogen atom is revolving in anticlockwise direction in a circular orbit of radius R (see figure). (a) Obtain an expression for the orbital magnetic dipole

In the Bohr model of the hydrogen atom, the electron circuulates around the nucleus in a path of radius 5xx10^

In a hydrogen atom, the binding energy of the electron in the ground state is E1. Then the frequency of revolution of nth electron in the nth orbits is

quantum mechanics - Ignoring spin, what is its orbital magnetic moment of an electron in a hydrogen atom in the 2p orbital? - Physics Stack Exchange
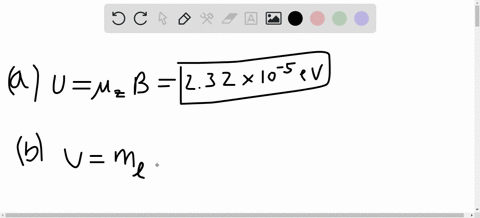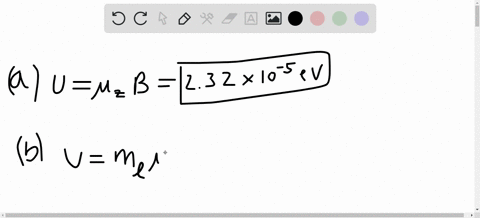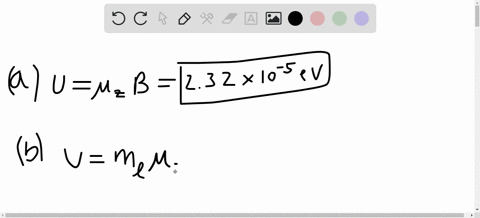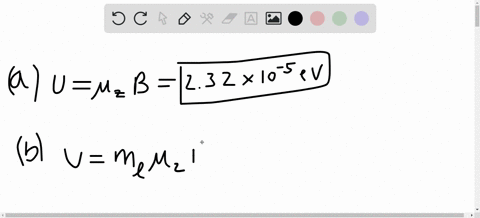
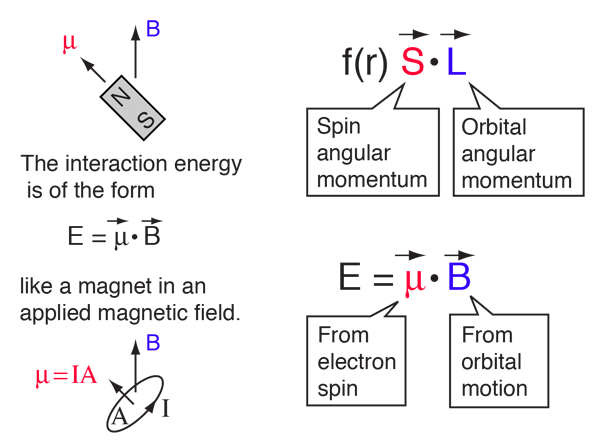
SOLVED:A hydrogen atom in a 3p state is placed in a uniform external magnetic field B . Consider the interaction of the magnetic field with the atom's orbital magnetic dipole moment. (a)
The minimum magnetic dipole moment of electron in hydrogen atom is - Sarthaks eConnect | Largest Online Education Community
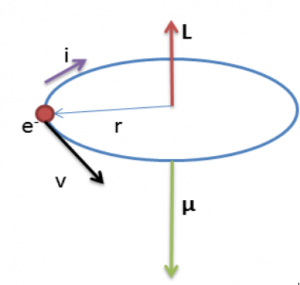
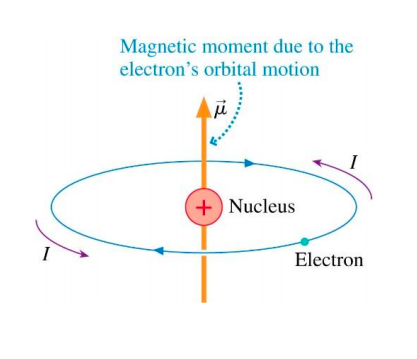
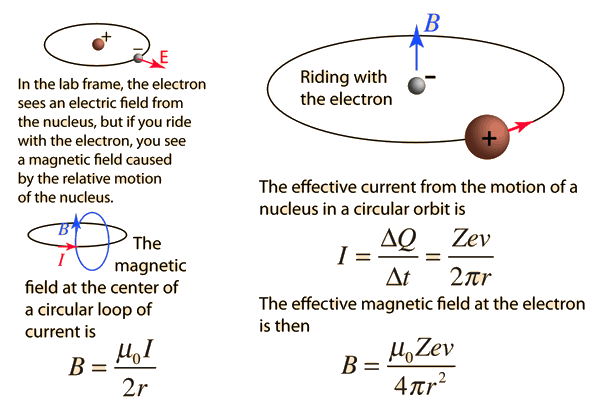
Define the term magnetic moment of current loop. Derive the expression forthe magnetic moment when an electron revolves at a speed 'v' around an orbitof radius r in hydrogen atom. Also calculate

42. The ratio of magnetic dipole moment of an electron of charge e and mass m in Bohr's orbit in hydrogen atom to its angular momentum is :